
R-Biopharm
Next-generation TB diagnostics, molecular STI testing, food safety, and clinical solutions for the Caribbean
R-Biopharm is a global leader in diagnostic innovation. Their IP-10 tuberculosis assays (RIDASCREEN TB and RIDA QUICK TB) deliver higher sensitivity than the gold-standard QuantiFERON — 85.2% vs 71.7% in active TB, and 91.4% vs 74.3% in high-incidence HIV+ populations. This makes them the critical TB diagnostic upgrade for Caribbean Ministries of Health and PAHO programs. Their STI portfolio — TandemPlex multiplex PCR panels, Vivalytic POC molecular, and RIDA GENE real-time PCR — is purpose-built for high-prevalence regions. With food safety ELISA and allergen testing completing the portfolio, R-Biopharm is a comprehensive diagnostics partner for the Caribbean.
30
Products
9
Product Families
6
Sectors Served
Product Portfolio
Tuberculosis — IP-10 Release Assays
Next-generation TB diagnostics using IP-10 (CXCL10) biomarker instead of IFN-γ. IP-10 is released at 100x the concentration of interferon-gamma, enabling superior sensitivity especially in immunocompromised patients. CE IVDR certified. The only viable competitor to QuantiFERON-TB Gold Plus, with proven higher sensitivity in multicentric clinical studies. Three-component system: RIDA TB Tubes for blood collection/stimulation, then choose RIDASCREEN ELISA (high-volume) or RIDA QUICK lateral flow (rapid/POC) for IP-10 detection.

RIDA TB Tubes
Three-tube blood collection system for IP-10 induction. Negative Control, TB-specific Test Tube (M. tuberculosis antigens), and Positive Control. Draw 1 mL blood per tube, incubate 16–24h at 37°C, collect plasma. Compatible with both RIDASCREEN and RIDA QUICK downstream detection.
- 3 tubes per patient (N/T/P)
- 1 mL blood per tube
- 16–24h incubation at 37°C
- Ambient shipping
RIDASCREEN TB (ELISA)
Quantitative ELISA for IP-10 measurement in stimulated plasma. 96-well microplate processes 28 patients per plate (3 wells each). Automatable on DSX, Agility, or any open ELISA platform. CE IVDR multicentric validation: 85.2% sensitivity, 90.3% specificity in active TB. 91.4% sensitivity in high-incidence HIV+ cohort vs 74.3% for QuantiFERON.
- 28 patients per 96-well plate
- 85.2% sensitivity (active TB)
- 91.4% sensitivity (HIV+ cohort)
- Automated ELISA compatible
RIDA QUICK TB (Lateral Flow)
Lateral flow cassette for IP-10 detection in stimulated plasma. One cassette per sample (3 per patient). Results read by RIDA Q3 reader. Ideal for clinics, border health, and field screening. 80.0% sensitivity, 91.3% specificity. 92.1% positive agreement with RIDASCREEN TB.
- Lateral flow rapid format
- 80.0% sensitivity, 91.3% specificity
- RIDA Q3 reader quantification
- Clinic and field-ready
RIDA Q3 Reader
Compact lateral flow reader with integrated software for RIDA QUICK TB cassette analysis. Automated measurement, calculation, and result documentation. Quality-assured, audit-ready results.
- Automated LF reading
- Integrated result calculation
- Audit-ready documentation
- Compact benchtop format
STI & Sexual Health — TandemPlex Multiplex PCR
TandemPlex panels use proprietary multiplex tandem PCR (MT-PCR) technology — a two-step nested PCR that pre-amplifies targets before detection, achieving maximum sensitivity and specificity even in highly multiplexed panels. Detect viruses, bacteria, parasites, and fungi in a single run. Critical for Caribbean labs where co-infections are common and comprehensive syndromic testing saves lives.
TandemPlex Urinogenital & Resistance 12-well
The flagship STI panel: detects Chlamydia trachomatis (including LGV), Neisseria gonorrhoeae (dual-target), Trichomonas vaginalis, Mycoplasma genitalium, plus conditionally pathogenic flora (M. hominis, U. parvum, U. urealyticum). Includes ceftriaxone resistance for N. gonorrhoeae, fluoroquinolone resistance for M. genitalium, and macrolide resistance for M. genitalium — guiding targeted antibiotic therapy.
- 9 STI pathogens + 3 resistance markers
- Dual-target N. gonorrhoeae
- Antibiotic resistance detection
- Swab, urine, CSF, plasma
TandemPlex Urinogenital 8-well
Core STI screening panel: Chlamydia trachomatis, Neisseria gonorrhoeae (dual-target), Trichomonas vaginalis, Mycoplasma genitalium, plus M. hominis, U. parvum, and U. urealyticum. Ideal for routine screening in clinics and public health programs.
- 7 STI pathogens
- Core CT/NG/TV/MG panel
- Ureaplasma species included
- High-throughput compatible
TandemPlex STI 16-well
Expanded STI screening covering Chlamydia, Mycoplasma genitalium, Neisseria gonorrhoeae (dual-target), Trichomonas vaginalis, plus M. hominis, Streptococcus agalactiae (GBS), U. urealyticum, and U. parvum. Designed for comprehensive prenatal and sexual health screening.
- 8 pathogens in one run
- Includes GBS for prenatal
- Comprehensive screening
- Semi-automated workflow
TandemPlex HPV High-Low Risk 16-well
Full HPV genotyping panel detecting 20 HPV types: 15 high-risk (HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, 68, 82) and 5 low-risk (HPV 6, 11, 42, 43, 44). Individual genotype identification enables risk stratification and cervical cancer screening programs.
- 20 HPV genotypes
- 15 high-risk + 5 low-risk
- Individual type ID
- Cervical cancer screening
TandemPlex Viral & Syphilis 12-well
Viral STI panel: HSV-1, HSV-2, VZV, EBV, CMV, HHV-6, plus Treponema pallidum (syphilis), Enterovirus, Parechovirus, and Adenovirus. Essential for lesion/ulcer workup and congenital infection screening.
- 10 targets including syphilis
- HSV-1/HSV-2 differentiation
- Herpes family coverage
- Lesion/ulcer diagnostics
TandemPlex Vaginitis & Vaginosis 12-well
Comprehensive vaginitis panel: Bacterial vaginosis (Gardnerella vaginalis, Atopobium vaginae), Lactobacillus flora analysis (L. crispatus, L. iners, L. jensenii, L. gasseri), Trichomonas vaginalis, and Candida species (C. albicans, C. tropicalis, C. glabrata, C. krusei, C. parapsilosis). Automatic vaginal flora scoring.
- BV + Candida + Trichomonas
- Lactobacillus flora profiling
- Automatic BV scoring
- No extraction needed (partial)
TandemPlex Lesions & Ulcers 12-well
Targeted panel for genital ulcer disease: HSV-1, HSV-2, Treponema pallidum (syphilis), Haemophilus ducreyi (chancroid), Chlamydia trachomatis LGV, and Mpox. Critical for differential diagnosis of genital lesions in tropical settings.
- Genital ulcer differential
- Syphilis + chancroid + LGV
- HSV typing
- Mpox detection
STI & Sexual Health — RIDA GENE Real-Time PCR
RIDA GENE kits use standard real-time PCR for flexible, targeted STI detection. Compatible with any extraction system and most PCR platforms. All kits can be combined in workflows for customized testing. 100 reactions per kit with 20 freeze/thaw cycles for cost-effective use.
RIDA GENE STI Mycoplasma Panel
Multiplex real-time PCR for Mycoplasma hominis, Mycoplasma genitalium, Ureaplasma urealyticum, and Ureaplasma parvum from genital swabs and urine.
- 4 Mycoplasma/Ureaplasma targets
- Swab or urine samples
- 100 reactions per kit
- Open platform compatible
RIDA GENE Mpox Virus
Real-time PCR for Mpox virus (MPXV) DNA detection from swab samples. Detects both Clade I and Clade II variants — critical given the 2024-2025 Mpox emergence in Caribbean travel networks.
- Clade I + Clade II detection
- Swab samples
- Combinable with STI panels
- Rapid turnaround
STI Point-of-Care — Vivalytic / RIDA Care
Vivalytic (RIDA Care) is a fully automated all-in-one molecular diagnostic platform developed by Bosch Healthcare Solutions. Sample-to-result in a single cartridge — no extraction, no pipetting, no technical expertise required. Ideal for Caribbean clinics, urgent care, and sexual health clinics that need molecular-grade results without a centralized lab.
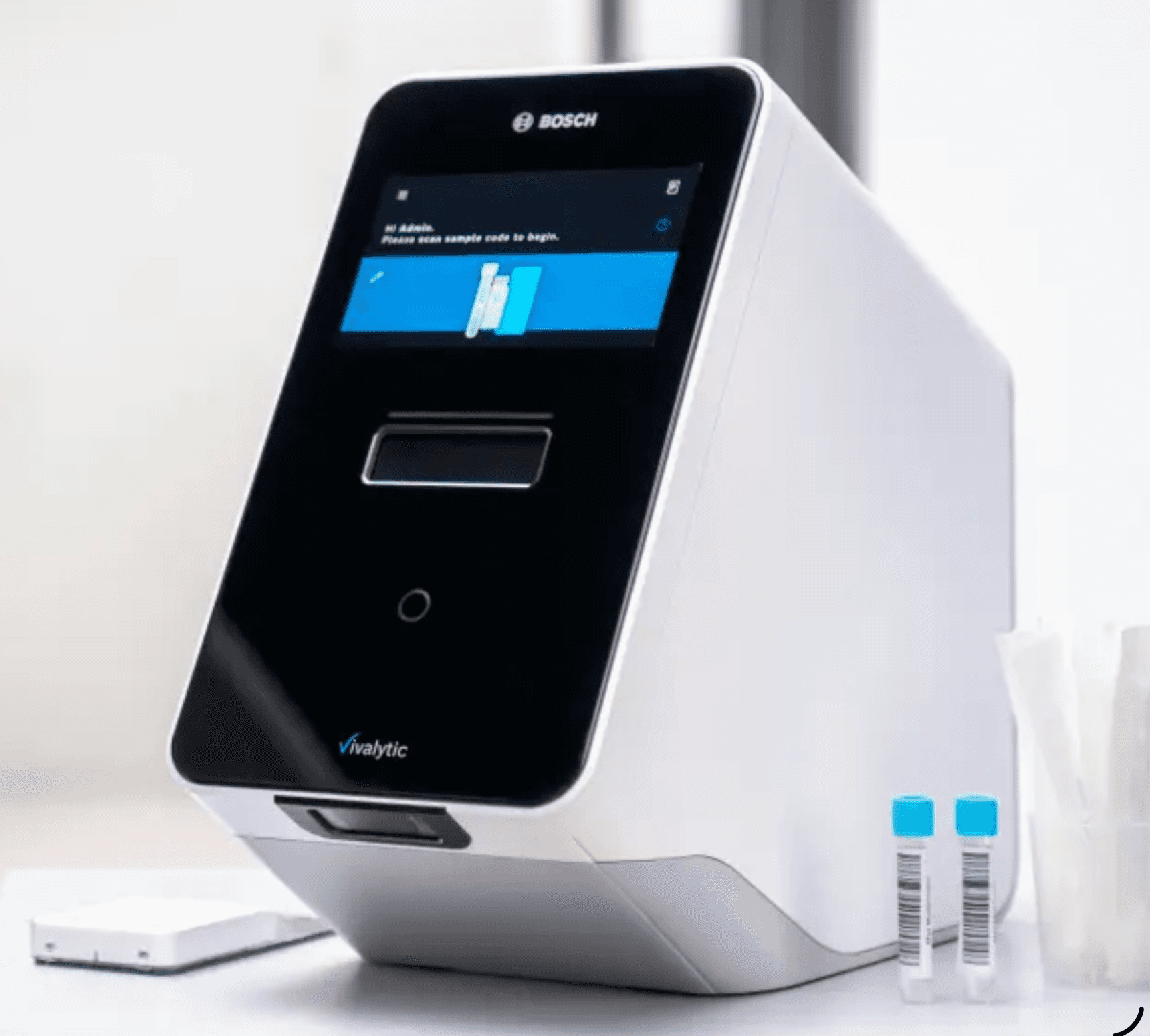
Vivalytic STI Panel
Fully automated detection of 10 sexually transmitted pathogens in one cartridge: N. gonorrhoeae, M. genitalium, U. urealyticum, C. trachomatis, T. vaginalis, H. ducreyi, M. hominis, T. pallidum, HSV-1, and HSV-2. Walk-away operation from various swab types and urine.
- 10 STI pathogens per cartridge
- Fully automated sample-to-result
- No extraction needed
- Swab or urine input
Vivalytic Mycoplasma Panel
Point-of-care detection of Mycoplasma genitalium, Mycoplasma hominis, and Ureaplasma parvum/urealyticum in a single automated cartridge.
- 3 targets per cartridge
- Fully automated
- Low sample throughput ideal
- Clinic-ready operation
Vivalytic Mpox Panel
Point-of-care molecular detection of MPXV virus and Orthopoxvirus species. Detects Clade 1 and Clade 2. Cartridge-based, fully automated, results at bedside.
- MPXV + Orthopoxvirus
- Clade 1 + Clade 2
- Fully automated cartridge
- No lab infrastructure needed
Quality Controls — Microbix QAPs
External quality controls for STI, respiratory, and GI molecular assays. Manufactured by Microbix Biosystems, distributed through R-Biopharm. Non-infectious, clinically relevant matrix, cross-platform compatible.
PROCEEDx STI Quality Controls
External quality assessment panels for STI molecular assays. Non-infectious native pathogen matrix, compatible across instruments and assay platforms. Essential for CAP/COLA accreditation.
- Non-infectious matrix
- Cross-platform compatible
- 2-year shelf life
- POC testing compatible
Clinical Diagnostics
ELISA and rapid tests for infectious disease and autoimmune diagnostics.
RIDASCREEN ELISA Kits
Comprehensive ELISA panel for infectious diseases: norovirus, rotavirus, adenovirus, C. difficile, H. pylori, and parasitology.
- 96-well microplate
- Infectious disease panel
- GI pathogen detection
- Batch processing
RIDA QUICK Rapid Tests
Lateral flow rapid tests for norovirus, rotavirus, influenza A/B, RSV, Strep A, and other acute infections.
- Lateral flow format
- 10-15 min results
- POC suitable
- Respiratory & GI panels
Clinical Allergy Diagnostics
Comprehensive allergy testing platform for IgE-mediated (Type I) and IgG-mediated (Type III) allergies. RIDA qLine immunoblot panels for allergen-specific IgE detection across food, aeroallergen, and insect venom categories. RIDASCREEN Spec. IgG Foodscreen ELISA for delayed food allergy profiling with up to 300+ antigens. Automated on RIDA qLine autoBlot (36 membranes/run) and standard ELISA platforms.
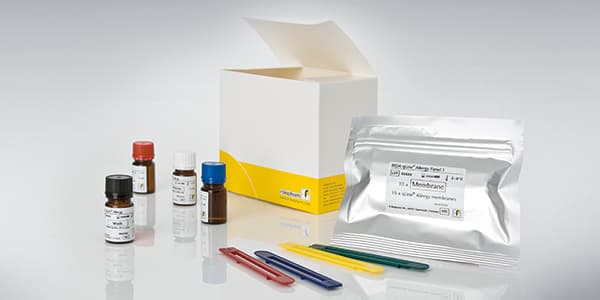
RIDA qLine Allergy Panels 1-4
Quantitative allergen-specific IgE immunoblot on nitrocellulose membranes. Four panels covering inhalant allergens (grasses, trees, weeds, mites, molds, animal epithelia), food allergens (milk, egg, wheat, soy, peanut, tree nuts, fish, shellfish), and insect venoms. Automated processing on RIDA qLine autoBlot. Results read by RIDA qLine Scan with RIDA qLine SOFT software for standardized, audit-ready reporting.
- 4 panels (inhalant, food, mixed, pediatric)
- Quantitative IgE results
- RIDA qLine autoBlot automated
- CE/ISO certified
RIDASCREEN Spec. IgG Foodscreen
Modular ELISA system for quantitative detection of food-specific IgG antibodies. Prescreen plate tests 22 common foods across 4 patients. Extended plates cover 90-91 antigens each, with customized plates offering 300+ food antigen selections. Identifies delayed-type (Type III) food allergies occurring 12-48 hours after exposure. 24-month shelf life, calibrated against international reference standards.
- Up to 300+ food antigens
- Modular plate system
- Prescreen + extended panels
- 24-month shelf life
RIDA CHIP FoodGuide
Miniaturized microarray enzyme immunoassay (M-EIA) testing up to 211 food antigens in just two wells. High-density allergen profiling with minimal serum volume. Available in 25, 50, 100, and 200 allergen configurations for flexible food allergy screening.
- Up to 211 food antigens
- Microarray format
- Minimal serum volume
- Flexible configurations

RIDA qLine autoBlot
Automated immunoblot processing system handling up to 36 membranes per run. Standardized incubation, washing, and staining for reproducible IgE and IgG allergy results. Paired with RIDA qLine Scan reader and RIDA qLine SOFT analysis software for complete walkaway allergy diagnostics.
- 36 membranes per run
- Fully automated processing
- RIDA qLine Scan reader
- SOFT analysis software
Food Safety & Allergen Testing
ELISA, rapid, and PCR-based tests for food allergens, mycotoxins, and pathogens — relevant for Caribbean food import testing and hotel/resort food safety programs.
RIDASCREEN Food Allergen ELISA
Quantitative ELISA for gluten, peanut, soy, milk, egg, almond, hazelnut, and other food allergens. AOAC-approved methods.
- AOAC approved
- Quantitative results
- Major allergens covered
- HACCP validation
Mycotoxin Testing
ELISA and lateral flow tests for aflatoxin, ochratoxin, DON, fumonisin, and zearalenone in grains, spices, and feed.
- Aflatoxin/DON/OTA
- ELISA & rapid formats
- Grain/spice/feed matrices
- Regulatory compliant
Instruments
From the RIDA Xplore semi-automated multiplex PCR to the Vivalytic POC molecular platform, RIDA UNITY full automation, ELISA systems, and rapid test readers. A complete instrument portfolio for every lab size.

RIDA Xplore System
Next-generation semi-automated MT-PCR platform. Detects up to 40 target genes per run across up to 24 samples. Less than 3.5 hours total turnaround (1h55m preamplification + 1h15m PCR). Approximately 72 samples/day across 3 runs. UV deck sterilization, snap-in reagents, automatic results calling. Pairs with RIDA Cycler 384. Under 1m² bench space. Compatible with all TandemPlex STI, respiratory, and GI panels.
- Up to 40 targets per run
- 24 samples/run, ~72/day
- < 3.5h turnaround, 3 min setup
- UV sterilization, < 1m² footprint

Vivalytic / RIDA Care Analyzer
Fully automated all-in-one molecular diagnostic platform by Bosch Healthcare Solutions. Touchscreen operation, barcode scanning, cartridge-based sample-to-result PCR. No extraction, no pipetting. STI, Mycoplasma, Mpox, respiratory, and TB cartridges available. Ideal for clinics, urgent care, and satellite labs without molecular expertise.
- Fully automated cartridge PCR
- Touchscreen with barcode scan
- No extraction or pipetting
- Multiple test menus available
RIDA UNITY Automated PCR
Fully automated real-time PCR system for medium-to-high-volume molecular diagnostics. Walk-away from extraction through results.
- Fully automated
- Extraction through PCR
- Walk-away operation
- RIDA GENE kit compatible
Agility ELISA System
12-plate fully automated ELISA system for high-throughput food safety and clinical immunoassay testing.
- 12-plate capacity
- Fully automated
- High throughput
- Open platform
RIDA QUICK SCAN II
Lateral flow reader for quantitative RIDA QUICK rapid tests. Quality-assured documentation and secure result analysis.
- Quantitative reading
- QA documentation
- Result archiving
- Multi-test compatible
Applications
- Tuberculosis screening (latent and active TB)
- Immigration and border health TB screening
- HIV co-infection TB detection
- Occupational health TB programs
- STI diagnostics
- Food allergen testing
- Mycotoxin screening
- Clinical GI pathogen diagnosis
- IgG food allergy profiling
- Food safety / HACCP
Sectors Served
Interested in R-Biopharm?
Contact us for pricing, product availability, and technical support for R-Biopharm products in your territory.
